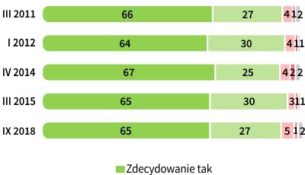
Determining The Empirical Formula Of Magnesium Oxide.
Magnesium Oxide Experiment Lab Report Essay Objective: One objective was to figure out if the burnt Mg ashes weigh more than the product which is Magnesium Metal. Another objective was determining the formula of the compound that results when Magnesium and Oxygen react.In the Lab Determining the Empirical Formula of Magnesium Oxide, students set out to find if there is a true 1:1 ratio in the empirical formula of MgO. This was determined by burning the Magnesium until a white smoke started to protrude. This showed the reaction of Oxygen combining with Magnesium to form Magnesium Oxide.Lab Determining the Empirical Formula of Magnesium Oxide Abstract In the Lab Determining the Empirical Formula of Magnesium Oxide, students set out to find if there is a true 1:1 ratio in the empirical formula of MgO. This was determined by burning the Magnesium until a white smoke started to protrude.
Empirical Formula for the Compound using Whole Numbers Mg5O3 Mg14O13. DISCUSSION In order to have magnesium oxide, MgO, the magnesium strip had to be heated. Under normal circumstances, room temperature, magnesium metal, Mg, reacts very slowly with the oxygen, O, in the air.Conclusion In our experiment we gathered the data of Magnesium reactions of being ignited to determine the simplest formula of magnesium oxide. The mass of product to be used in all following calculation is based on the final mass of the crucible minus the mass of the empty crucible, therefore the mass of product is .416 g.

Determining the empirical formula of magnesium oxide lab Essay - Major Essays Since multiplying the ratio by five gives the closest values to whole numbers, therefore the empirical formula of the oxide is Mg6O5. The third trial was not taken into account because the mass after combustion was not recorded.